- Folder Technical News
- Views 1513
- Last Updated 02/04/2025
Sorbitol is a natural polyol widely used in various fields. However, the use of products containing sorbitol must comply with relevant regulations.
1. What is Sorbitol?
Sorbitol is a type of polyol (a naturally occurring sweetener in the sugar alcohol group), with the chemical formula C₆H₁₄O₆. It is naturally present in many fruits such as apples, pears, peaches, and plums, and can also be industrially synthesized from glucose through a hydrogenation process.
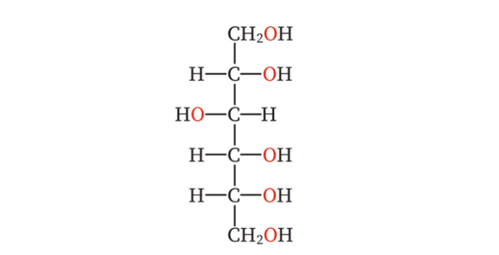
Figure 1. Structural formula of Sorbitol
In terms of sensory characteristics, sorbitol appears as a colorless crystalline solid or a clear syrup, odorless, with a mild, cooling sweetness. Its sweetness is only 50-60% of sucrose (table sugar) but does not cause tooth decay and has a low glycemic index (GI), making it suitable for people with diabetes or those who need to control their sugar intake.
2. Applications of Sorbitol
In the food industry, sorbitol is commonly used as a sugar substitute, especially in confectionery production, to reduce calorie intake and minimize the risk of tooth decay. Additionally, it has moisturizing and glazing properties, helping to maintain freshness and improve the texture of food products.
In the medical and pharmaceutical fields, sorbitol is used as an osmotic laxative to treat constipation, increasing osmotic pressure in the intestines and promoting hydration, thereby aiding digestion. Furthermore, sorbitol serves as an excipient in the production of vitamin C-containing medicines and personal care products (such as toothpaste) due to its moisture-retaining and stabilizing properties.
3. Undesirable Effects of Use
Sorbitol may cause some undesirable effects, particularly on the digestive and metabolic systems. Regarding the endocrine and metabolic effects, sorbitol can lead to water and electrolyte imbalance and increase the risk of lactic acidosis. In the digestive system, common side effects include diarrhea, abdominal pain, nausea, and vomiting, especially in individuals with irritable bowel syndrome or bloating. Therefore, caution should be exercised when using sorbitol in sensitive individuals to avoid these adverse effects.
4. Current Regulations
According to the European Food Safety Authority (EFSA), sorbitol intake should not exceed 20 g/day for children and 50 g/day for adults to avoid adverse health effects.
To ensure consumer awareness, food labels containing more than 10% polyols (authorized under Annex II of Regulation (EC) No. 1333/2008) must include the mandatory warning: "Excessive consumption may cause laxative effects," in accordance with Annex III of Regulation (EU) No. 1169/2011 of the European Parliament and the Council.
In Vietnam, sorbitol is a common food additive and is permitted for use in certain food products according to QCVN 4-8:2010/BYT. Sorbitol has an "unlimited" ADI (Acceptable Daily Intake), meaning there is no specified upper limit for its acceptable daily consumption.
5. Usage Recommendations
For consumers: Carefully read product labels to check sorbitol content and follow usage instructions. Sorbitol should not be used long-term as a laxative but only as a supportive product alongside dietary treatment. When sorbitol is used in combination with activated charcoal, it may increase blood sodium levels in both adults and children. Therefore, water and electrolyte balance should be monitored, and the lowest possible dose of sorbitol should be used.
Caution should be exercised when using sorbitol in individuals with megacolon or irritable bowel syndrome.
For manufacturers: Ensure compliance with food additive labeling regulations, providing clear information about sorbitol content to help consumers make informed and safe choices.
Author
Vu Thi Nhat Le – Department of Nutrition and Food Additives
National Institute for Food Control
References
1. Vietnam National Pharmacopoeia, 2022, Sorbitol
2. Panoff, L. (2020) What is Sorbitol? benefits, uses, side effects, and more, Healthline. Available at: https://www.healthline.com/nutrition/what-is-sorbitol#storage-handling (Accessed: 22 March 2025).
3. Regulation - 1333/2008 - en - additives - EUR-lex (no date) EUR. Available at: https://eur-lex.europa.eu/eli/reg/2008/1333/oj (Accessed: 22 March 2025).
4. Scientific opinion on the substantiation of health claims related to the sugar replacers xylitol, sorbitol, mannitol, Maltitol, lactitol, Isomalt, erythritol, d‐tagatose, isomaltulose, sucralose and polydextrose and maintenance of tooth mineralisation by decreasing tooth demineralisation (ID 463, 464, 563, 618, 647, 1182, 1591, 2907, 2921, 4300), and reduction of post‐prandial glycaemic responses (ID 617, 619, 669, 1590, 1762, 2903, 2908, 2920) pursuant to Article 13(1) of regulation (EC) no 1924/2006 -. Available at: https://efsa.onlinelibrary.wiley.com/doi/10.2903/j.efsa.2011.2076 (Accessed: 22 March 2025).
5. Circular 24/2019/TT-BYT on the management and use of food additives
6. QCVN 4-8:2010/BYT - National Technical Regulation on Food Additives – Synthetic Sweeteners